Ethyl (R)-(+)-4-Chloro-3-Hydroxybutyrate
: Overview and Applications
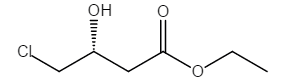
Ethyl (R)-(+)-4-Chloro-3-Hydroxybutyrate is a chemical compound with significant applications in pharmaceutical synthesis and organic chemistry. Below is a detailed overview of its properties, synthesis methods, and applications based on the search results provided.
1. Chemical Properties
Ethyl (R)-(+)-4-Chloro-3-Hydroxybutyrate is a colorless, transparent liquid at room temperature with a distinctive ester-like aroma. It exhibits moderate solubility in water and is miscible with common organic solvents.
2. Synthesis Methods:Ethyl (R)-(+)-4-Chloro-3-Hydroxybutyrate
2.1 Biocatalytic Synthesis
The compound can be synthesized through biocatalytic methods, such as the asymmetric reduction of ethyl 4-chloroacetoacetate. This process often employs genetically engineered E. coli strains that co-express aldehyde reductase and glucose dehydrogenase, enabling efficient coenzyme regeneration and high enantiomeric excess (ee) of the product. Optimal reaction conditions include a temperature of 32°C, beyond which the yield decreases.
2.2 Enzymatic Resolution:Ethyl (R)-(+)-4-Chloro-3-Hydroxybutyrate
Another approach involves the enzymatic resolution of racemic 4-chloro-3-hydroxybutyrate using lipases. For instance, Novozym 435 lipase in dioxane solvent has been shown to achieve an ee of over 99% under optimized conditions (30°C, 0.5 mol/L substrate concentration, 10 mg/mL enzyme concentration, and 35 hours reaction time).
2.3 Iodination Reaction
Ethyl (R)-(+)-4-Chloro-3-Hydroxybutyrate can also undergo iodination reactions. For example, reacting it with sodium iodide in acetone under reflux conditions for two days yields a high-purity product (99%) after purification via silica gel column chromatography.
3. Applications
3.1 Pharmaceutical Intermediate
This compound is a key intermediate in the synthesis of atorvastatin calcium, a widely used cholesterol-lowering drug.
3.2 Organic Synthesis:Ethyl (R)-(+)-4-Chloro-3-Hydroxybutyrate
Its chiral structure makes it valuable in asymmetric synthesis and the production of fine chemicals.
4. Key Research and References
- Biocatalytic Synthesis: Studies on the use of recombinant E. coli for asymmetric reduction and enzymatic resolution using lipases have been documented.
- Iodination Reaction: Detailed procedures for the iodination of Ethyl (R)-(+)-4-Chloro-3-Hydroxybutyrate are available in the literature.
Conclusion
Ethyl (R)-(+)-4-Chloro-3-Hydroxybutyrate is a versatile compound with significant applications in pharmaceutical and organic chemistry. Its synthesis via biocatalytic and enzymatic methods highlights its importance in green chemistry and sustainable production processes. For further details, refer to the original sources.
Company: Hangzhou Fengjing Biotechnology Co., Ltd.
Email: hwl5726@163.com
Address: No. 8 Zhongtong Road, Eshan She Ethnic Township, Tonglu County, Zhejiang Province